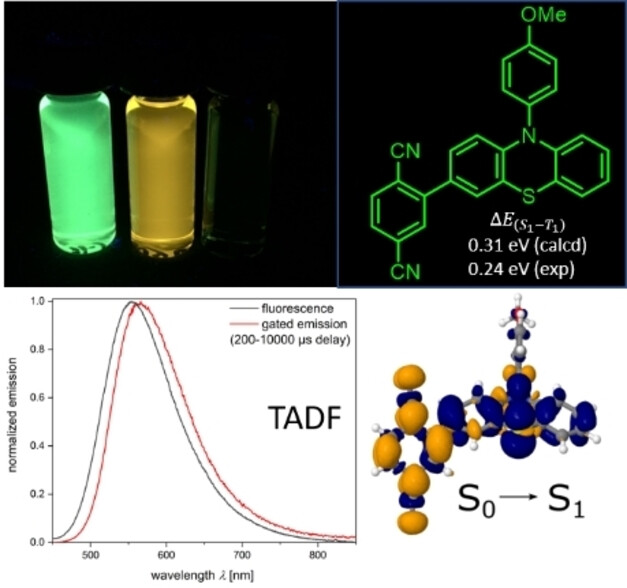
The bromine-lithium exchange-borylation-Suzuki sequence efficiently furnishes phenothiazine-terephthalonitrile donor-acceptor dyads and triads in high yields. In contrast to most phenothiazine-acceptor conjugates the title compounds are ligated in p-position to the phenothiazine nitrogen atom. Moreover, the acceptors are either directly linked or ligated by an arylene bridge and p-anisyl N-substituents on the phenothiazine are chosen to lock the tricycle into an intra-configuration. Cyclic voltammetry reveals effects of bridging and ligation of the N-substituent. Optical spectroscopy likewise displays similar band gaps, large Stokes shifts and substantial to high quantum yields in solution, in the solid state and in PMMA matrix. Time-resolved fluorescence spectroscopy indicates quite long fluorescence decay times in solution and emission components in the microsecond time range. TADF properties are further assessed by fluorescence increase in deoxygenated solution, gated emission spectroscopy and temperature-dependent determination of phosphorescence. The nature of the electronically excited states is investigated by DFT/MRCI. While for the directly ligated dyad a singlet-triplet energy gap ΔE(S1-T1) of 0.24 eV can be estimated and is consistently confirmed by quantum chemical calculations on the lowest energy conformer, even lower ΔE(S1-T1) of 0.029 and 0.008 eV are estimated for the investigated dyads and the triad in the solid state and in PMMA matrix.
Read more in:
3-Ligated Phenothiazinyl-terephthalonitrile Dyads and Triads - Synthesis, Electronic Properties, Delayed Fluorescence and Electronic Structure
Dr. Laura N. Kloeters, Jan A. Meissner, Lysander Presser, Prof. Dr. Markus Suta, Prof. Dr. Jan Meisner, Prof. Dr. Thomas J.J. Müller
Chem. Eur. J., 29, 2023, e202301114