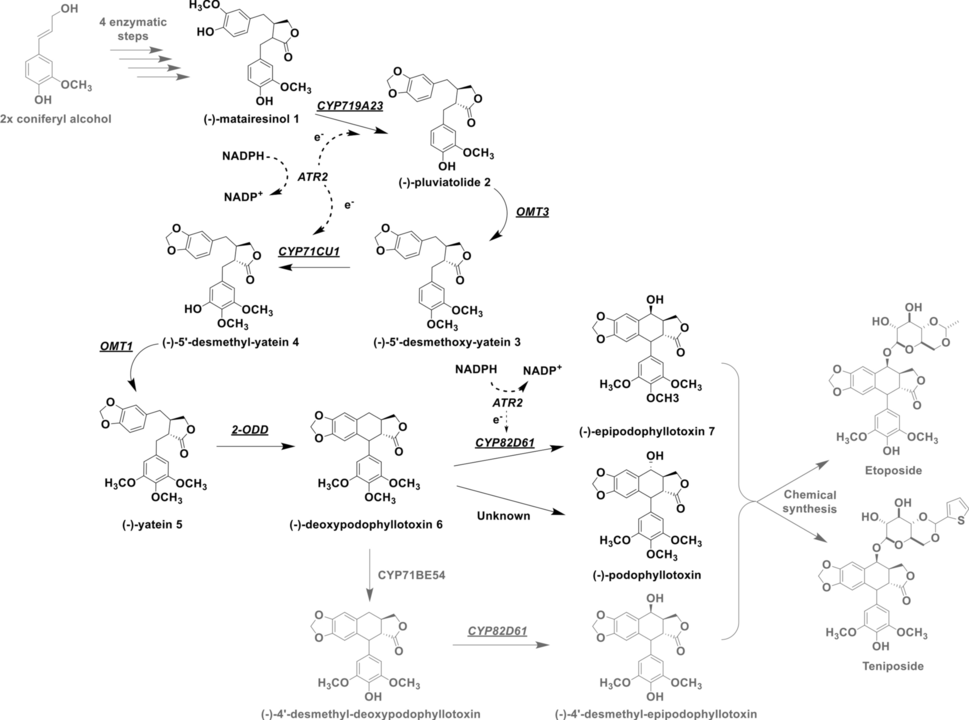
Davide Decembrino and colleagues from our group successfully established the synthesis of (−)−deoxypodophyllotoxin and (−)−epipodophyllotoxin, two direct precursors to the chemotherapeutics etoposide and teniposide, via a 6-step multi-enzyme cascade in E. coli. One of their major successes within these study was that two plant cytochrome P450 monooxygenases were functionally expressed in E. coli for the first time. The results recently appeared in Microbial Cell Factories.
"The aryltetralin lignan (-)-podophyllotoxin is a potent antiviral and anti-neoplastic compound that is mainly found in Podophyllum and Sinopodophyllum plant species. Over the years, the commercial demand for this compound rose notably because of the high clinical importance of its semi-synthetic chemotherapeutic derivatives etoposide and teniposide. To satisfy this demand, (-)-podophyllotoxin is conventionally isolated from the roots and rhizomes of Podophyllum hexandrum, which can only grow in few regions and is now endangered by overexploitation and environmental damage. For these reasons, targeting the biosynthesis of (-)-podophyllotoxin precursors or analogues is fundamental for the development of novel, more sustainable supply routes. In this study, a five-step multi-enzyme biotransformation of (-)-matairesinol to (-)-deoxypodophyllotoxin was established and proven effective with 98% yield (78.1 mg/L). Furthermore, the extension of this cascade to a sixth step leading to (-)-epipodophyllotoxin was evaluated. To this end, seven enzymes were combined in the reconstituted pathway involving inter alia three plant cytochrome P450 monooxygenases, with two of them being functionally expressed in E. coli for the first time."
Decembrino D, Raffaele A, Knöfel R, Girhard M, Urlacher V, 2021, Synthesis of (-)-deoxypodophyllotoxin and (-)-epipodophyllotoxin via a multi-enzyme cascade in E. coli, Microbial Cell Factories, 20:183, https://doi.org/10.1186/s12934-021-01673-5